A recent interviewee on Del Bigtree’s Highwire lost my attention when she referred to Koch’s Postulates as ‘old science’. In my simple way of thinking there is good science and bad science, not old and new science.
A while back I promised to write about the method we developed to visualise aluminium in biological tissues. To shine a light, pun intended, upon aluminium-selective fluorescence microscopy.
An understanding of how this method works gives significant insight into the interpretation of the images it produces. One such image is shown below.
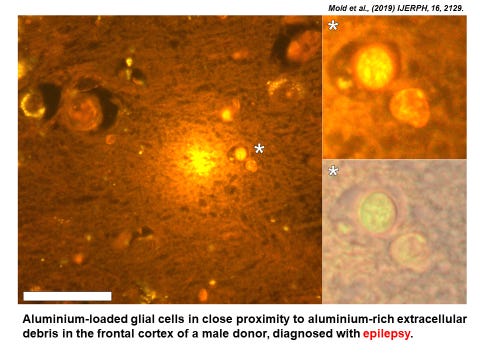
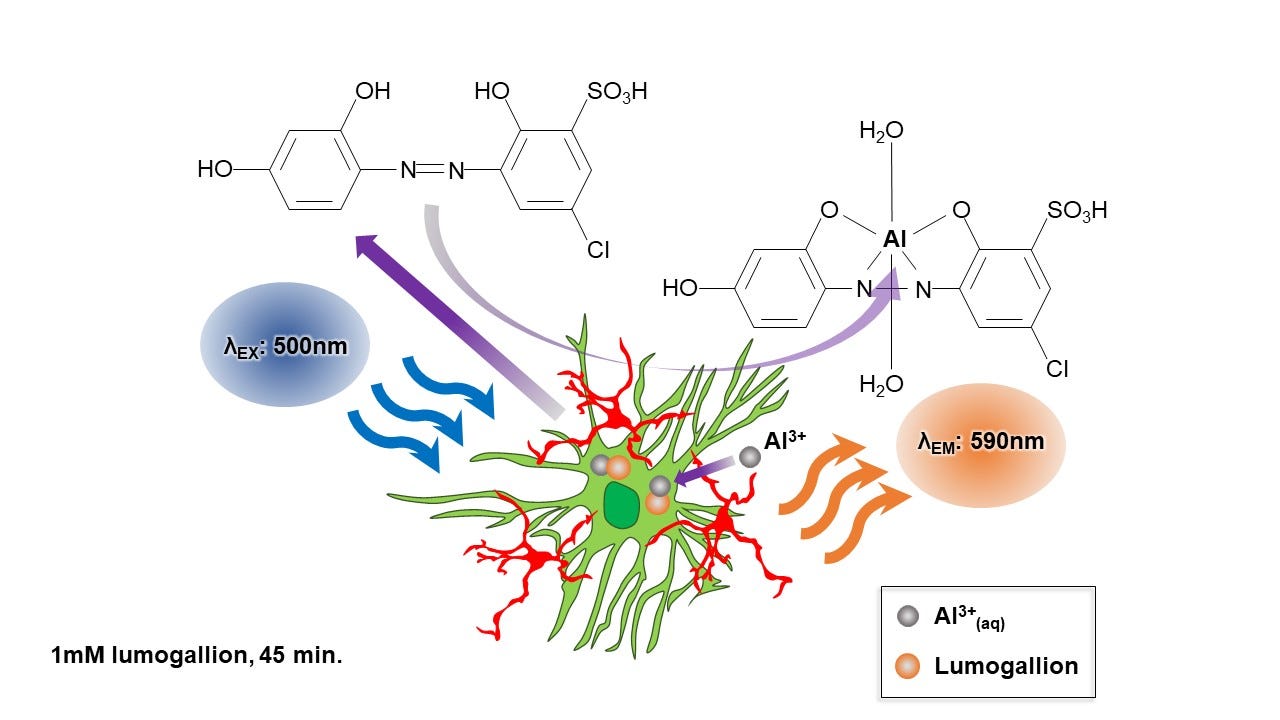
You can go to the original paper to see the figure in better detail. The image is an example of aluminium-selective fluorescence. Bright yellow/orange light identifies the presence of aluminium. Tissue sections, in this case brain tissue from a donor with epilepsy, are processed in the presence of a molecule called lumogallion. The schematic below shows how lumogallion combines with aluminium to produce characteristic fluorescence.
The figure shows that lumogallion, see its structure on the left, binds aluminium, see the new structure of the lumo-Al complex on the right. When the new lumo-Al complex is excited by light of a specific wavelength (500nm) it responds by emitting light at a new wavelength (590nm) and one that gives an aluminium-specific yellow/orange light. It is very important to note from this figure that lumogallion binds Al3+ , the free metal cation, to form the lumo-Al complex.
So, how does this required knowledge enable us to understand better the Al-specific fluorescence shown in the previous figure and again below.
For one it tells us that the aluminium-rich extracellular debris at the centre of the image is a rich source of Al3+ , it is not simply aluminium bound up in cellular, probably neuronal, debris. It is telling us that this aluminium is biologically available and therefore toxic to surrounding tissues. Now take a close look at the glial cells shown magnified in the inserts. The aluminium is seen as punctate deposits within the cytoplasm (cell body) of the glial cells. These ‘deposits’ are almost certainly membrane-bound as the glial cells have taken up the aluminium and secured it in vesicles. However, the fact that we can see the aluminium as bright yellow fluorescence tells us that some of the aluminium in these vesicles is in the form of the free metal cation, Al3+ . This is not unexpected as vesicles used to compartmentalise unwanted substances (the glial cells are the housekeeping cells of the brain) actively acidify their contents and in this case inadvertently solubilising the aluminium contained within them. The resulting Al3+ is toxic to the glial cell and will eventually cause its death adding further to the aluminium-rich extracellular debris.
Hopefully you can see now how each of these images showing the presence of aluminium tells a story about what was happening in situ, in this example, in brain tissue in someone who died with a diagnosis of adult onset epilepsy. Fluorescence microscopy of this ilk is ‘old’ science and I would say good science. The method was first developed by my PhD student Ambreen Mirza. Ambreen showed that only aluminium produced this characteristic fluorescence and that while lumogallion bound other metals including for example iron, the resulting lumo-metal complexes were silent with respect to fluorescence. However, the use of lumogallion to visualise aluminium in biology and especially human brain tissue was taken to an extraordinary new level by my colleague Dr Matt Mold and he has been responsible for myriad wonderful images published in many of our papers. He is to be congratulated for the very best good (old) science.
PS Many thanks to those who have chosen to support this substack financially. While my substack will always remain free to all your contributions are extremely helpful, for example in maintaining our website where you can find a lifetime’s work and many of Dr Mold’s wonderful images of aluminium.



Absolutely FASCINATING information! Thank you so much for sharing! After reading about the aluminum sulfate being accidentally dumped in the drinking water, I am very happy that I have been using only RO treated drinking water since 1989. Now that I have been reading your posts, it is very obvious that the "vaccine" manufacturers do not want the general public to be aware that the jabs contain aluminum which is neurotoxic. I recently did some research on ingredients in "vaccines" used for veterinary applications. Many still contain thimerosal and most contain aluminum compounds. Thimerosol is still being designated as a "preservative". I always forward your posts to my NMD.
100%. I heard that as well and was not impressed by that comment about “old science”. Thank you for all your hard work and excellent explanations.