Recent email correspondence with an aluminium evangelist raised the thorny issue of what makes, for example, a metal, like aluminium, essential to life. She had been taken to task by a journalist who said that the fact that tea plants accumulate aluminium and may even grow better in the presence of aluminium proved that aluminium was an essential element for tea. This particular line of thinking, and especially that presence must infer some sort of requirement, takes me back to those halcyon days when the aluminium industry actually engaged in discussions on aluminium. They would often attempt to counter any suggestion that aluminium might be toxic in humans by pointing out that since aluminium is omnipresent throughout the human body it must be helpful in some way. The inference being that the body would not function in the absence of aluminium. Similar suggestions as to the benign if not essential nature of aluminium have been touted more recently by eminent vaccine developers such as Paul Offit (Children’s Hospital of Philadelphia, CHOP) and Andrew Pollard (Oxford). The latter choosing to use the vernacular of the ‘yellow polka dot bikini’ (itsy bitsy teeny weeny) to describe aluminium adjuvants in vaccines while Offit would tell any parent who would listen that aluminium was essential to their child’s health. He even published this advice on CHOP’s website, though this critical information seems to have been removed more recently. How lucky we are to have such paediatricians as custodians of children’s health and well being. I am digressing.
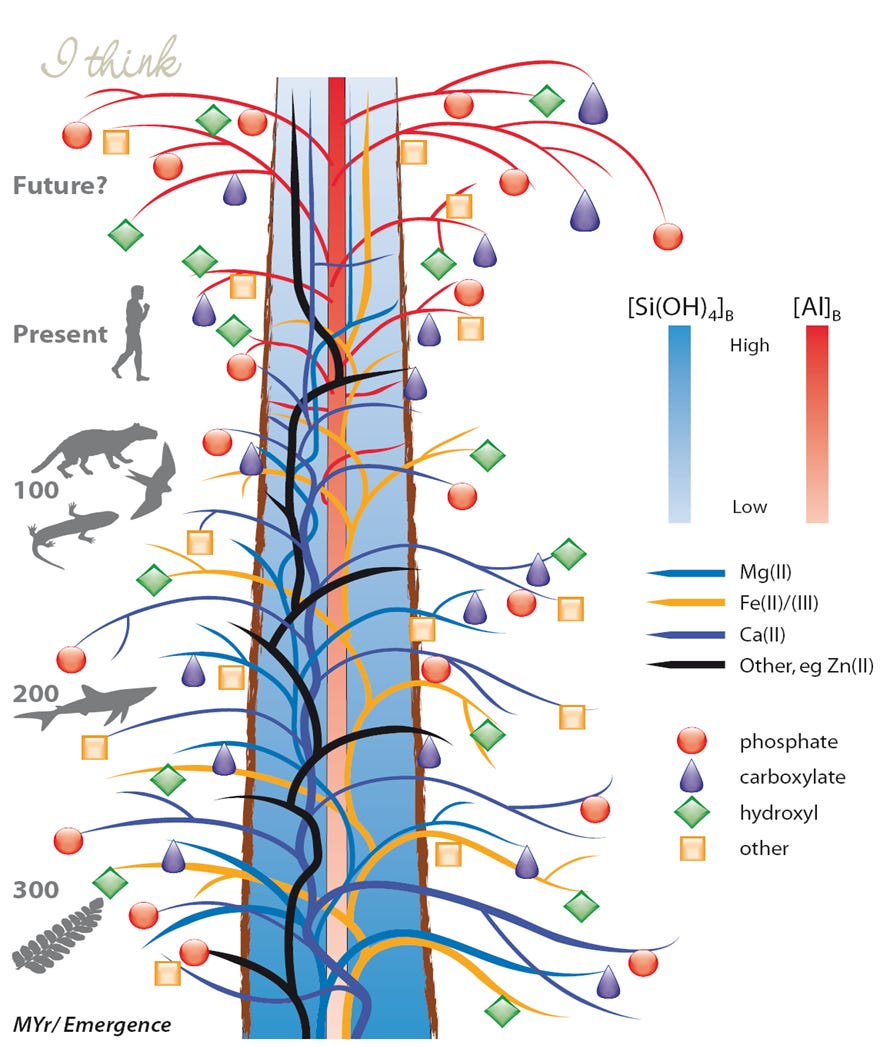
I wrote a paper on the non-essentiality of aluminium and silicon and since that paper was also supporting Darwin’s 200th birthday I included my version of his tree of life.
Please refer to the original paper for an in depth explanation of the figure. However, its take home message is simply that silicon is non-essential because of a complete lack of appropriate biochemistry while aluminium is non-essential because of a lack of biological availability. My tree of life predicts that with the advent of the aluminium age comes increased biological availability of aluminium and commensurately increased toxicity.
The critical aspect to understand about aluminium is not a lack of biological reactivity, quite the opposite is the case, but that wherever and whenever that aluminium interacts with extant biochemistry it is only disruptive. I have written about this extensively, a recent example being. I have also tried to summarise this in the below image, taken from, in which the various ways that aluminium might cross a cell membrane are summarised. Again, please refer to the original paper for a full explanation. If you do happen to be challenged by someone on this subject then ask them for an example of a trivalent metal ( a metal ion carrying three positive charges) that is involved in essential biochemistry. If they try to trick you by suggesting iron (Fe3+) then make sure to tell them that this form of iron is only stored and not used in iron biochemistry per se.
So, I hear some exasperated voices asking, what about tea plants and aluminium. Well the brown tea referred to quite correctly by the aforementioned journalist was brought to India (and elsewhere) by the British to avoid having to buy tea from China. Tea plants were grown intensively and in areas that could not sustain such agricultural practice. Soils became acidified and edaphic sinks of previously biologically unavailable aluminium became available and were taken up by the tea plants. Some plants survived and even continued to flourish under the acidic aluminium-replete conditions. These plants had an extensive polyphenol biochemistry and these compounds, the same ones that give tea its distinctive colour and flavour, bind aluminium with high avidity, essentially locking it up in the tissues, the tea leaves. Inherent biochemistry selected during the evolution of the species for a function distinct from anything to do with aluminium becomes a natural protection against biologically available aluminium. Note that these same tea plants grow perfectly well in the complete absence of aluminium. The observation that tea plants may actually grow better in the presence of aluminium is explained by the fact that soil acidity is toxic to tea plants and where aluminium is also present the acidity is countered by the amphoteric nature of biologically available aluminium. It is why we use aluminium salts as antacids. I will write about other examples of this relationship between aluminium and acidity in forthcoming substacks.
So, the journalist was at least half correct. The moral of this aluminium story must be that what appears to be the case is not exactly what it seems and only an understanding of the underlying biology in the context of Darwinian natural selection can explain the bare essentials of life.



I have been joining battle on twitter. Two scientists both tried the 'aluminum is ubiquitous' dismissal. Well yes, but bound by silica. So, in a reversal of a common trope I tell them that suggesting aluminum is safe because it's abundant is like saying that chlorine gas is safe because salt is abundant. I don't think either of them enjoyed that at all.
No-one has mentioned tea to me, but surely one could easily counter with the fact that there are plants that can handle soil with all kinds of metal pollution? I wish that my discussions could be so genteel.
So...it's still true that a little bit of knowledge is a dangerous thing?! ;-)