While I no longer have a laboratory and the opportunity to take the science of human exposure to aluminium forward I do feel obliged to use this medium, Dr’s Newsletter, to put you in touch with the latest research in the field. Of course, this process will not be altogether benign, you will have to suffer my opinions on the new research and therein my interpretation of its importance.
Today, I want to bring to your attention a recent review on aluminium in food. You can use the following link to download a pdf of the new paper. I am aware that aluminium in food is a subject that interests many of you since I receive may queries on this subject both through this substack but also by direct email. However, before you get too much embroiled in the data presented in this paper I want to give you some context. Some background as to what takes place in your gut when you ingest aluminium through your diet.
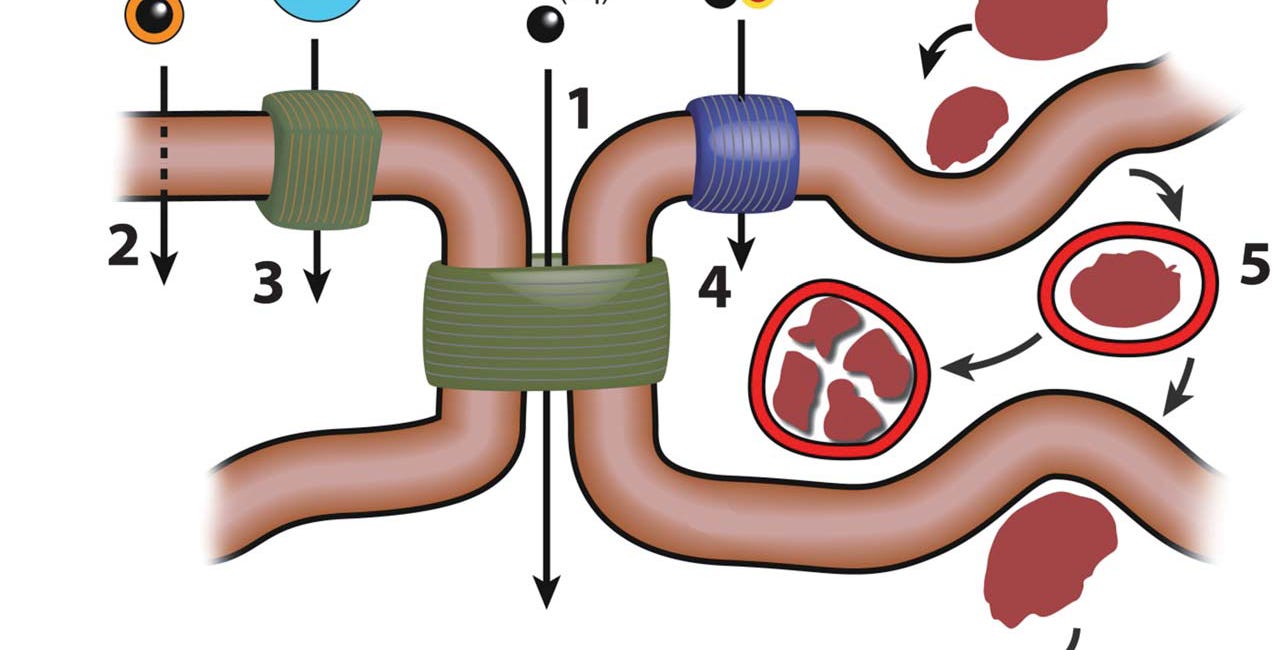
As you know, digestion begins in the mouth which through the secretion of saliva is a fairly circumneutral environment. This means that unless the ingested food is naturally quite acidic any aluminium contained within it will be in a largely insoluble form. The mouth prepares the ingested food for delivery to the strongly acidic environment of the stomach. This is where digestion begins in earnest and it is also the one part of the gut where aluminium previously in an insoluble form may be solubilised even to the extent that aluminium may be present in the stomach as its free metal cation Al3+. An intact fully functioning stomach is not where products of digestion including aluminium are absorbed into the bloodstream. The stomach prepares food for further digestion and subsequent absorption of essential nutrients in the remainder of the gastrointestinal tract, the small and large intestines. It also, subconsciously, assuming a conscious stomach, prepares aluminium for life in the remainder of the gut. This means that the form of aluminium in the stomach will influence whether or not it is available for absorption into the bloodstream via primarily the small intestine. The acidic environment of the stomach largely supports the presence of soluble aluminium in the form of its free metal cation. However, other constituents of food that have been released in the stomach may bind Al3+ to form small soluble complexes of aluminium. Such constituents might include small organic acids, natural ingredients of many foods, as well as other less natural constituents such as fluoride, see my previous posts on this subject, and glyphosate, the toxicity of which is a somewhat controversial subject.
I have written about glyphosate and human toxicity in my book. As a biologist with a strong background in chemistry I have struggled to understand why glyphosate might be toxic in humans and I have come to the conclusion that its main role in the human body is to facilitate the absorption of aluminium from the gut. Like fluoride, glyphosate binds aluminium with some avidity in the acidic environment of the stomach. This means that when the aluminium-glyphosate complex enters the less acidic environment of the small intestine it does so in a form that promotes the movement of aluminium across the intestinal membrane into the blood. To appreciate the mechanism involved in the movement of aluminium across biological membranes please refer to the information in a previous substack.
Stable, small complexes of aluminium such as those formed with fluoride and glyphosate do not hydrolyse upon entering the circumneutral environment of the duodenum and ileum. This means that they remain as small soluble complexes and unlike, for example, Al3+, they will not immediately precipitate as much less soluble forms such as aluminium hydroxide and aluminium hydroxyphosphate.
You will understand from this brief discussion that it is not just about how much aluminium is present in food, though this is important, but what accompanies aluminium in the gut and specifically in the acidic environment of the stomach where the assumption is often made that aluminium will not form soluble complexes but only be present as its free metal cation. This is a subject I cover in some depth in my book and it remains a subject that suffers from a lack of high quality published science. The proportion of ingested aluminium that is absorbed into the bloodstream may be anything from 1-30%, we simply do not know. However, you can be absolutely certain that if your ingested food is accompanied by constituents such as fluoride and glyphosate then the amount of aluminium becoming systemic and gaining entry to vital organs such as the heart and brain will be towards the higher estimate of 30%.
Food glorious food, yes, but better served as a dish without aluminium.
Dr’s Newsletter is free for all to read and comment upon. My objective being to spread the gospel according to Mr Aluminium as widely as possible. However, I am thankful to those few who believe my words are worth the odd dollar and choose to take out a paid subscription. You are keeping me on my toes and eager to share what little knowledge I have on my favourite subject, aluminium.


I watched your 2018 interview with Del Bigtree yesterday. The information you shared was absolutely fascinating! I forwarded the link to about 20 people including my NMD. I am really hoping that everyone I shared the link with will watch it because this is most likely one of the most important interviews I have ever watched.
This past week, I re-read Dr. Dale Bredesen's book on Alzheimer's Disease, I was shocked to see that he discusses toxins in the brain but does not consider aluminum an issue. He made a comment that aluminum has not been proven to cause or be associated with AD! I think he needs to read your book and your Dr's Newsletter.
Thanks for sharing more excellent information!
This is so important...The word has to get out about this...I personally don't eat a lot of processed food, but some for sure (more than I'd like), and I've been rolling it back further since first reading your work on food and aluminum...Thank you.