In a recent substack, It Started with a Fish (I hope that like me you have been singing this song (Hot Chocolate) ever since.), I promised to write about our research on aluminium and mucus.
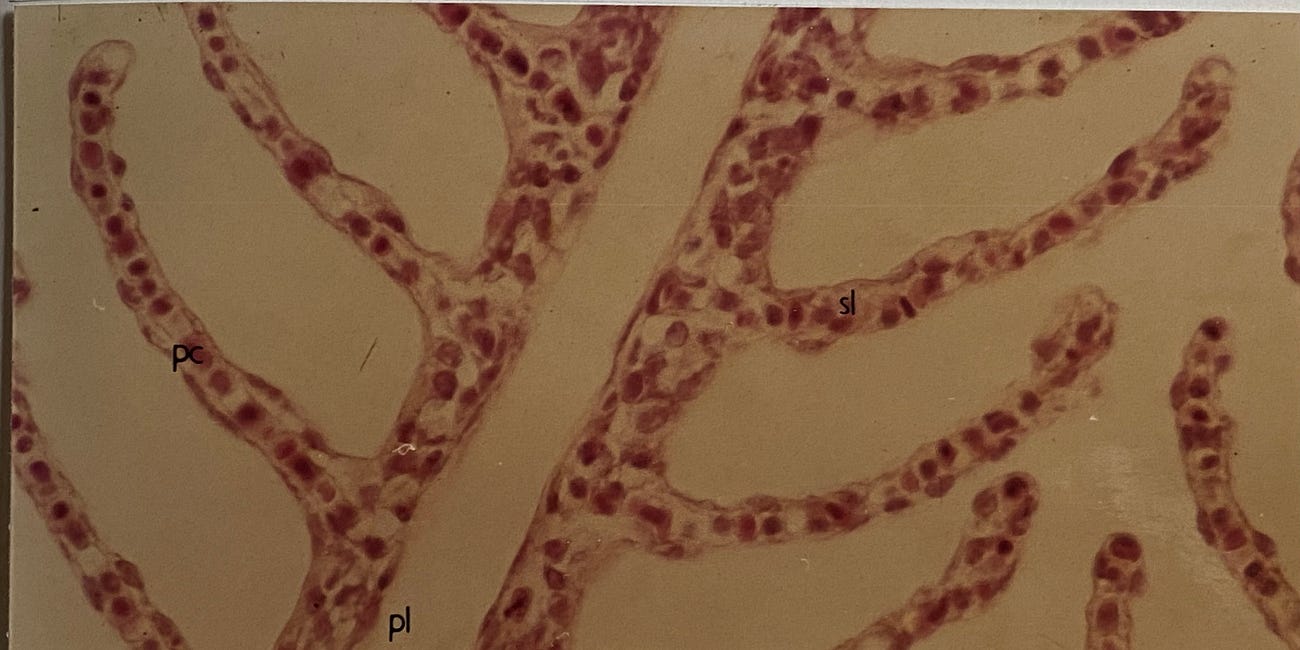
Mucus may not be everyone’s cup of tea, it certainly has bad press, but it is keeping you alive each and every moment of the day. Exactly akin to the fish gill a steady stream of mucus passes over all of the ‘external’ internal surfaces of the body. From the dilute mucus that constitutes saliva in your mouth to its concentrated counterpart that protects the lining of the stomach from the acid it produces in digesting food. It protects the lung from the air that we breath and provides the lubrication essential to successful reproduction. Mucus has myriad functions in the human body, too many to write about here, and one can be certain that when a mucus layer is disrupted, as it is in acute aluminium toxicity in fish, trouble follows. The biochemistry of mucus evolved in the complete absence of biologically available aluminium and early in my scientific career I was determined to understand how it was impacted by aluminium.
I published one major paper specifically on this subject. Unfortunately it is not open access and so I will try to summarise its main findings here and you are more than welcome to drop me an email should you wish to have a pdf of the full paper. Mucus is produced by mucous cells situated in epi/endothelia. It is secreted as a concentrate and diluted (hydrated) as required at membrane surfaces. Its main component, excluding water, is the glycopeptide, mucin. Mucus is a highly conserved (evolutionarily successful) composite varying primarily only in its water content regardless of its location in the body. We prepared solutions of mucin and observed how aluminium influenced its properties.
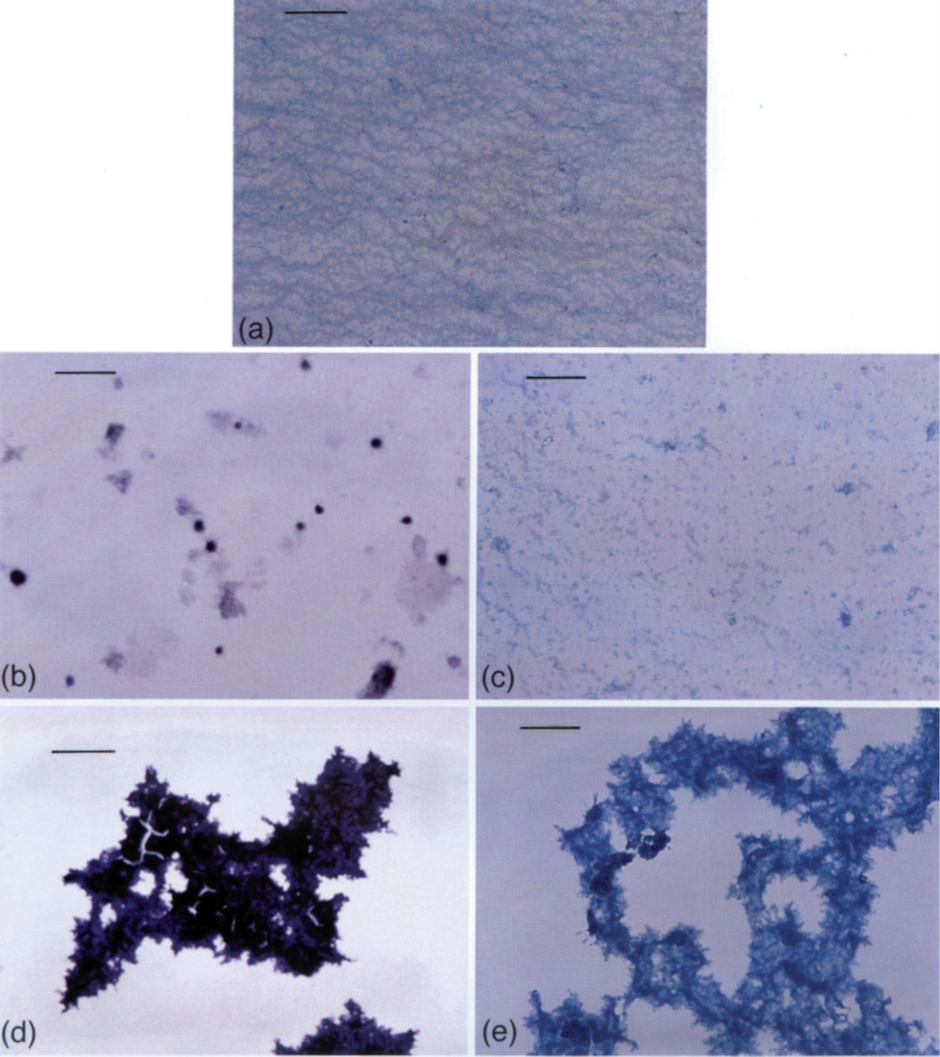
It came as no surprise, bearing in mind what we knew from fish, that high concentrations of aluminium caused the precipitation of mucin (mucus). However, what was much more interesting and perhaps more relevant to human physiology was what was happening to mucus prior to its precipitation. We found that prior to the precipitation of mucus by aluminium the viscosity of mucus increased significantly. The increase in viscosity was caused by mucin binding aluminium and the formation of distinct mucin-aluminium complexes. The latter took the form of spheres as shown in the figure below.
Image (a) uses the stain Alcian blue to show the structure of mucus in the absence of aluminium. The scale bar is 0.2mm. Notice a regular perhaps sponge-like structure. This structure is completely disrupted in the presence of aluminium, images (b) and (d), where the use of a modified haematoxylin stain identifies the presence of aluminium as a purple colour. Images (c) and (e) show how aluminium affects the structure of mucus using Alcian blue. Images (b) and (c) show the mucus structure prior to its precipitation and coincident with the increase in mucus viscosity. Images (d) and (e) show the structure of mucus upon its precipitation. At this point the viscosity of the mucus drops to something equivalent to the viscosity of water.
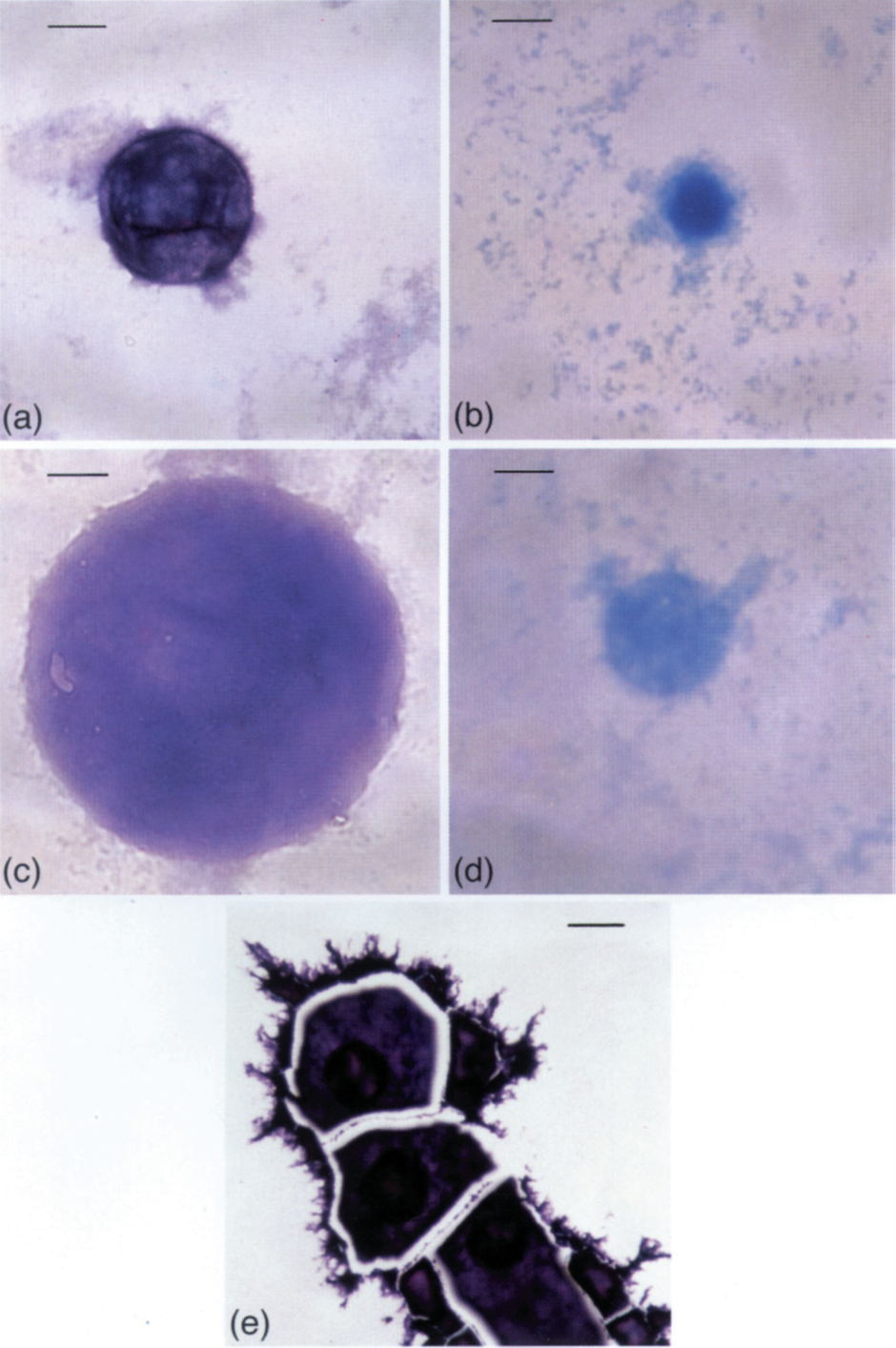
I was very interested by the formation of mucin-aluminium complexes and their apparent assembly into near-spherical particles. I was able to investigate these particles more closely as shown in the images below.
Image (a) and (c) show individual particles stained to show aluminium. The scale bar is 0.01mm. Image (b) and (d) show the exact same particles stained with Alcian blue to show the mucin glycopeptide. Note, as I did at the time, that these spherical particles, mucin-aluminium footballs, appear to have a soft centre. The orientation of the mucin-aluminium complexes is such that aluminium sits on the outside of the football and the mucin glycopeptide forms the inside of the football. I did attempt to prove this by sectioning particles and showing their cross section. I was able to do this but alas I did not publish these images and they are no longer available to me.
Image (e), scale bar 0.02mm, is important because it shows that the mucin-aluminium footballs act as nucleation sites for the subsequent precipitation of aluminium and mucin from solution. They act as catalysts of aggregation.
I love this research for a number of reasons. I presented it at several scientific meetings ranging from materials science to fish physiology. The research was well received. I believed at the time and continue to believe today that this research has important implications for human exposure to aluminium. The research demonstrated not only the potential destructive nature of aluminium-based antacids in the stomach but also how aluminium captured by the mucus lining the lung might influence the viscosity, the streaming, of these lung mucus layers. Any change in the viscosity of lung mucus is critical to its function as we see quite plainly in diseases such as cystic fibrosis. For me the story of mucus and aluminium started with a fish but it remains a trail least travelled in human physiology.


Given that aluminium sulfate is used around the world for clarifying water by flocculation of organic debris, and also as in astringent (to clot blood from small wounds), we might also expect that it would coagulate mucin protein. But you didn’t just assume this would happen. You proved that it does, including the thickening effect, and the potential destructive effect that this would have on the digestive system and the lungs etc.
So when we talk about "toxicity of ingested aluminium” it would be naive to just say “only 1% is absorbed through the gut”. We should also consider the destruction of the mucus within the gut and consequences of this weakened protection against many unwanted species.
Question: Is there a pathway for aluminium ions to enter the lungs and react with the mucus? (besides breathing airborne aluminium). I am particularly concerned about two grandchildren who live with cystic fibrosis.
Thank you
This is great information! Thank you for sharing!