I have heard it said many times that PhD’s are not what they used to be. I am old father William (to borrow from Lewis Carroll) and what I know for sure is that PhD research is hard and I worked very hard in achieving my PhD. I also enjoyed this period of my life immensely. My epiphany, and almost certainly the event that brought me to where I am now, happened late in the final months of my PhD research. I write, somewhat poetically about this moment in detail in my book. I will only precis that critical time herein.
I don’t actually remember if I was beginning to doubt my working hypothesis or perhaps my ability to implement the critical experiments but I do remember quite clearly the moment that science took hold of my life. On that particular early spring morning I experienced, quite literally, the sweet smell of success. Silicon had protected against acute toxicity of aluminium in fish. I was not the only one to rejoice this moment. My PhD supervisor, my ‘Father in Science’ JD Birchall OBE FRS rushed off a manuscript to Nature (well as rushed as is possible in the days before an effective internet). John Maddox, the editor of Nature at that time, was almost as eager and the paper was published within a few short weeks.
The research demonstrated that 0.1mmol/L silicon completely protected salmon parr from an acute exposure to aluminium. What a wonderful relief and how special it felt to defend my PhD thesis knowing that the main finding had already been published in Nature. What a cocky young scientist I almost certainly was at that time! However, all was not exactly as it seemed. Several years later I was leading a small group of scientists at Keele University and we had the opportunity to confirm the data published in the Nature paper. However, to my surprise, this time we were unable to show that 0.1mmol/L silicon (by then we understood the active form of silicon to be silicic acid) protected against acute aluminium toxicity in fish. What!?!
While alarm bells rang initially I also knew what I had found in my PhD research. What was the explanation? The thesis in question was that aluminium in the form of hydroxyaluminosilicates (HAS) (from the reaction of aluminium hydroxide with silicic acid) is non toxic. We knew from detailed chemistry experiments that I had carried out and published that HAS formed at the concentration of silicic acid (0.1mmol/L) used in the Nature research. So why did this concentration of silicic acid not protect against acute aluminium toxicity in the new experiments. The answer can be found in our understanding of acute aluminium toxicity in fish. I wrote about this in a previous substack.
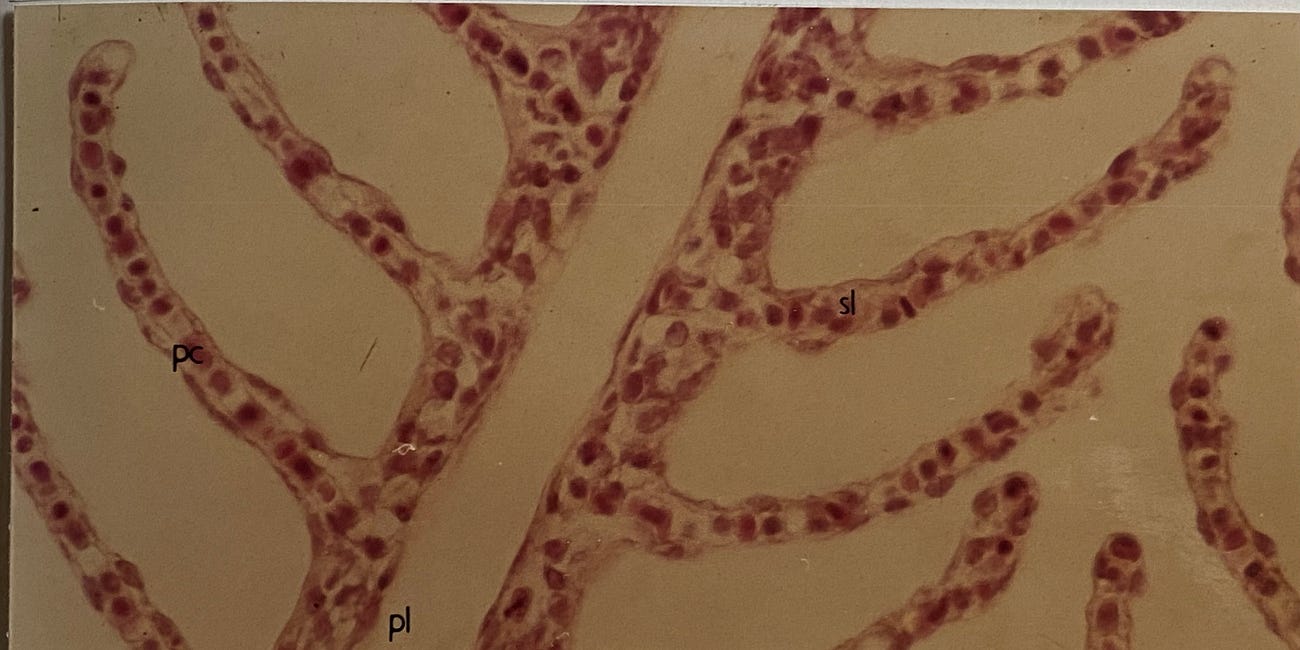
To be effective in preventing the toxicity of aluminium HAS must be stable at the surface of the fish gill. The HAS must not break down to release aluminium for binding at functional groups at the gill epithelium. What we discovered in our new research was that HAS formed in the presence of 0.1mmol/L silicic acid were not sufficiently stable to prevent their dissolution at the gill surface. For example, when HAS were formed in the presence of 1.0mmol/L silicic acid they completely protected against acute aluminium toxicity in fish. The HAS did not dissolve at the surface of the fish gill.
So, and here comes the serendipity bit, why did 0.1mmol/L silicic acid protect against acute aluminium toxicity in my PhD research. In fact it didn’t but a close examination of the experimental set-up used in my PhD research revealed the answer. In achieving the final experimental conditions to which fish were exposed, stock solutions of aluminium and silicic acid were pre-mixed and it was during this pre-mixing phase that HAS were formed. So, HAS formed, at for example 1.0mmol/L silicic acid when thereafter diluted into the experimental tanks containing 0.1mmol/L silicic acid remained stable for the 96h duration of the toxicity trial. I have probably lost a few of you by now through my clumsy description but if you are interested then do take a look at my PhD thesis where you will find both a diagram and a photograph of the experimental system. It should help you to understand what I am trying to explain.
The crux of this cautionary tale is that my PhD research should not really have been successful but I was not to know this at that time. It was our lack of understanding of the chemistry of the formation of HAS that allowed me then to design an experimental system that serendipitously resulted in a favourable outcome. Perhaps I was lucky or just perhaps luck knew that my working hypothesis was correct and that I just needed a guiding hand to bring this discovery to the world. I have always believed since that it has been my destiny to be the one who tells the story of human exposure to aluminium. I continue to tell this story here in my substack, old father William or not.


As a *former* MD-diagnosed Alzheimer’s sufferer, I am pleased to add my thanks and congratulations.
Several years ago, my MD-Administered MMSE 30 point “score” was in the mid-teens… beginning and supposedly irreversible Alzheimer’s. 😡
The doctors all told me there was no cure. But they were “working on it!”
So… in my first “overnighters” since University… I found your writings.
And I implemented the SILICA water regimen you recommended. A liter/quart of Fiji water daily.
My MMSE score last year was 28. This year… 29.
HIGH NORMAL!
At age 83, I still drive… safely… but not quite “brave” enough to get current with my Aircraft Pilot’s license. But brave enough to be heading to South America in a few weeks for trout fishing and then “hopping” over to South Africa for a safari!
All because of your research!
Thank you for your scholarship. Your findings on the damage of aluminum to the brain etc. is monumental! Now we must find more ways to remove it from our world.