I am of course grateful to all of those, all of you, that take the time to read my substack. I appreciate and it is often clear from your comments that many of you are coming to the subject of aluminium and specifically its toxicity for the first time. My assumptions otherwise, including that you may have read my book, are optimistic and so I decided I would share some general thoughts with you on why and how aluminium is toxic.
Let’s start at the beginning as I did nearly forty years ago and ask fish. Over twenty years ago I published some research that has proven in many ways to be revelatory in why aluminium is toxic. In a nutshell, though I think you can access the original paper here, I demonstrated that when aluminium was present in water as predominantly its free metal ion, Al3+, fish avoided very low concentrations of aluminium. They were able to detect and swim away from concentrations of aluminium as low as 27ppb. This is probably the approximate concentration of aluminium in potable drinking water. However, when aluminium was present in water as predominantly aluminium hydroxide, a solid phase of aluminium, fish did not avoid aluminium concentrations ten or even twenty times higher, for example, 500ppb. The fish are telling us that the biologically-reactive form of aluminium is Al3+ and it is the concentration of this form of aluminium that will determine its immediate toxicity. I write about this in some detail in my book but in brief, all forms of aluminium in an aqueous environment will dissolve to release Al3+. However, the rate at which this dissolution occurs will determine if toxicity ensues.
Those of you whom follow vaccine safety science will know that a form of aluminium hydroxide is used as an adjuvant in vaccines. Aluminium adjuvants are predominantly solids when they are injected in vaccines. The solid particles are the target of the ‘hungry’ immune-reactive cells that populate the vaccine injection site. They engulf the particles of aluminium adjuvant and store them in their cell bodies.
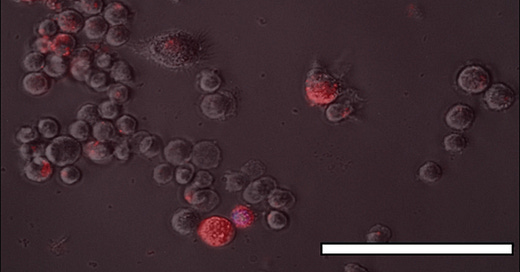
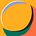
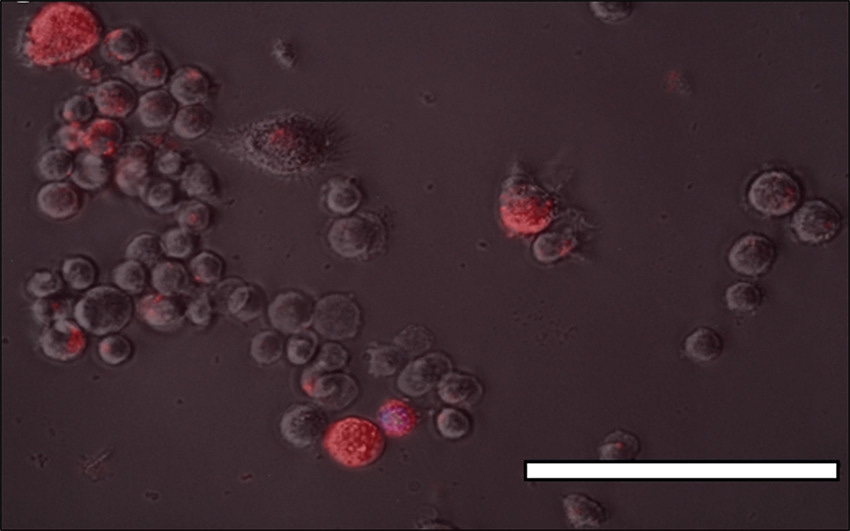
This image taken from one of our papers shows aluminium, fluorescent orange, inside macrophage cells. Similar evidence of the uptake of aluminium by monocytes typical of a vaccine injection site is available here. These cells are able to accumulate significant quantities of aluminium adjuvant without incurring immediate toxicity. This means that these migratory cells remain viable for days and perhaps longer during which time they can carry their cargo of toxic aluminium throughout the body. This transport of aluminium from a vaccine injection site to elsewhere in the body may be what we imaged in our research on aluminium in brain tissue in autism. See the image below taken from this paper.
Aluminium adjuvant in macrophages and other cells slowly dissolves to release burgeoning amounts of Al3+ and hence eventually kills the cell. This aluminium is then released into the tissue surrounding the dying cell where it may also bring about toxicity. This may be a mechanism whereby potentially toxic amounts of aluminium can be accumulated in, for example an infant’s brain tissue, in a relatively short period of time. A mechanism of aluminium-induced encephalopathy (accelerated cell death) similar to what is acknowledged to occur in dialysis encephalopathy.
So, Al3+ is the ‘why and how aluminium is toxic’ while all other forms of aluminium including aluminium adjuvants are the source of this biologically-reactive and hence toxic aluminium.


Thank you for covering this old but important ground.
This autumn see’s the introduction of Gardasil 9 to NHS vaccine schedule with double the aluminium of its predecessor Gardasil, this information is both relevant and necessary for full parental informed consent considering 31 lawsuits are currently underway Stateside for aluminium containing Gardasil vaccine harms.
At least the Biggest Medical Scandal of our time (according to the gender critical movement) is motivating concerned parents to 'do their own research' into the side effects of puberty blocking drugs, mutilating surgeries and pharma promoted ideology. Some of the TERFs I've peaked are starting to ask questions about vaccines too...